-
게시판-관련소식
뉴스레터 2024년 1월호
2024-01-30 by ISPE Korea
-
MFDS 소식
「의약품 제조 및 품질관리에 관한 규정」 고시전문
- 의약품 임상시험 계획승인에 관한 규정 일부개정고시
식품의약품안전처 고시 제2024-2호
의약품 임상시험 계획 승인에 관한 규정 일부개정 고시
1. 개정이유
임상시험계획 승인 제출자료 심사기준의 국제조화를 위해 최신 개발 시험법을 활용한 비임상시험자료의 제출 근거를 마련하는 등 규정을 개선하고자 함
-
-
GMP 가이드라인
-
[FDA] Policy for Testing of Alcohol (Ethanol) and Isopropyl Alcohol for Methanol
-
This guidance is intended to alert pharmaceutical manufacturers and pharmacists in Statelicensed pharmacies or Federal facilities who engage in drug compounding to the potential public health hazard of alcohol (ethyl alcohol or ethanol) or isopropyl alcohol contaminated with or substituted with methanol. During the Coronavirus Disease 2019 (COVID-19) public health emergency (PHE), the Food and Drug Administration (FDA) became aware of reports of fatal methanol poisoning of consumers who ingested alcohol-based hand sanitizer products that were manufactured with methanol or methanol-contaminated ethanol.
-
[PIC/S] Concept Paper on the revision of Annex 5 of the guideline on Good Manufacturing Practice for medicinal products - manufacture of immunological veterinary medicinal products
-
This concept paper addresses the need to update Annex 5 (manufacture of immunological veterinary medicinal products) of the Good Manufacturing Practice (GMP) guide. Annex 5 is common to the member states of the European Union (EU)/European Economic Area (EEA) as well as to the participating authorities of the Pharmaceutical Inspection Co-operation Scheme (PIC/S). The original version has not been revised since it was originally issued in 1992. Since that time, there has been extensive progress in the use of new technologies, significant changes in GMP following the adoption of the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) Q8, Q9, Q10 and Q11 guidelines,
-
-
ISPE 국내외 소식
교육안내
[3.22] GMP Engineering의 최신 Trend 및 그 실제
- 일시: 2024.03.22(금) 9:00~16:00
- 강사: 오형균(싸토리우스)
- 장소: 판교글로벌 R&D센터 A동 5층
- 대면(집합) 교육, 신청 접수중
[5.23] Extractables and Leachables Study
- 일시: 2024.05.23(목) 9:00~16:00
- 강사: 우성환, 강승훈(큐비디)
- 장소: 판교글로벌 R&D센터 A동 5층
- 대면(집합) 교육, 신청 접수중
[한글판] PHARMACEUTICAL ENGINEERING
January-February 2024 / Volume 44, Number 1
- QUALITY CONSIDERATIONS IN DISASTER RECOVERY: A Case Stidu
- New Good Practice Guide Covers Phamaceutical Gas Systems
- Cold Systems as a Solution to Decarbonize Water Purification
- Using Industry Survey Date to Shape Cell Therapy Facility Design
* 국제회원 및 교육회원에게 발송해 드립니다.
* ISPE KOREA 홈페이지-출판물-매거진에서도 확인하실수 있습니다.
ISPE KOREA 교육회원 모집
* 가입대상: 제약 및 Biopharma 관련업계 종사하는 모든 분
* 회원혜택
- 한글판 Pharmaceutical Engineering 열람 및 다운로드
- 온라인 & 오프라인 교육비 40% 할인
- 동영상 교육자료 무료시청
* 가입방법: 홈페이지(www.ispe.or.kr) - 회원가입 - 교육회원 - 신청서 작성 - 회원가입비 납부
* 회원가입비: 연 25만원
-
Upcoming Conferences
2024 ISPE Europe Annual Conference
16-18 April 2024
Lisbon, Portugal and Virtual
2024 ISPE China Annual Conference
12-13 April 2024
Hangzhou, Zhejiang, China
2024 ISPE Aseptic Conference
12-13 March 2024
Vienna, Austria and Virtual
2024 ISPE Facilities of the Future Conference
29-30 January 2024
San Francisco, CA USA and Virtual
-
Guidance Documents
Baseline Guide Vol 6: Biopharmaceutical Manufacturing Facilities 3rd Edition
Published: November 2023
Pages: 194
Member Price: $295.00
Non-Member Price: $595.00
Good Practice Guide: Process Gases 2nd Edition
Published: October 2023
Pages: 152
Member Price: $295.00
Non-Member Price: $595.00
Guide: 503B Compounding
Published: August 2023
Page: 122
Member Price: $295.00
Non-Member Price: $595.00
Good Practice Guide: Containment for Potent Compounds
Published: December 2022
Pages: 258
Member Price: $295.00
Non-Member price: $595.00
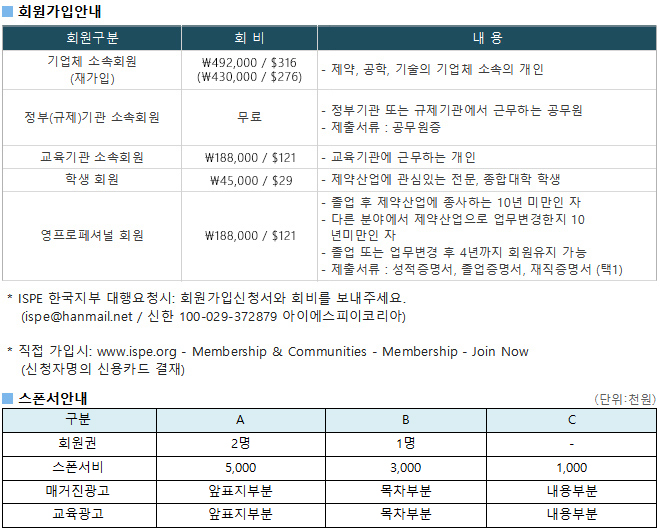
광고문의 TEL. 043-213-0442 E-MAIL. ispe@hanmail.net
ISPE Korea Affiliate | Office : 충북 청주시 흥덕구 직지대로 530, 702호 (송정동, 청주테크노S타워)
-
